Welcome to the Laboratory for cellular innate immunity
Our group performs translational immunological research studying compartment-specific characteristics of NK cells/ human innate lymphoid cells.

Our Research
Innate lymphoid cells (ILCs) are a family of innate immune cells with lymphoid phenotypes that lack rearranged antigen receptors. They are divided into three groups based on the expression of specific transcription factors, cell-surface markers, and signature cytokines. NK cells are a subset of ILC1s that play a critical role in innate immune surveillance and defense against viral infections and cancer.
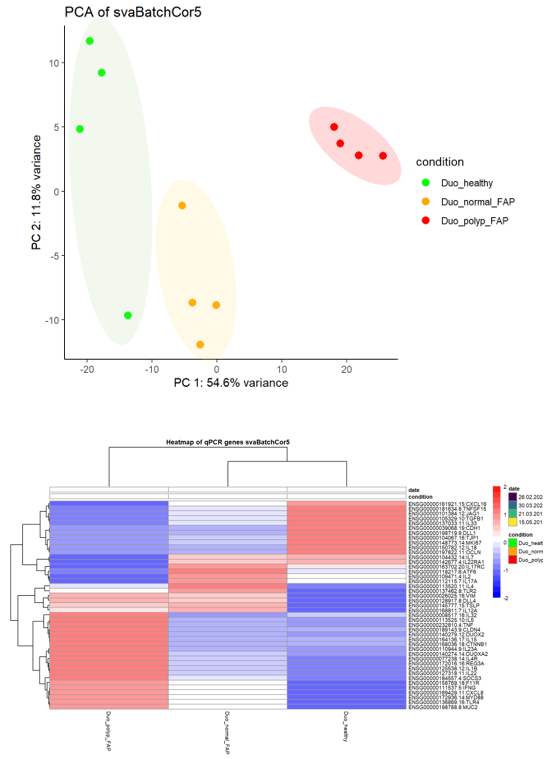
Our research aims to understand the compartment-specific physiological and pathological functions of human innate lymphocytes, including NK cells, for immune homeostasis, viral infections, and inflammatory diseases. Additionally, we investigate their role in hereditary gastrointestinal cancer syndromes, such as Lynch syndrome and familial adenomatous polyposis, to identify potential therapeutic targets for these diseases.
ILCs, including NK cells, play a significant role in maintaining intestinal health by promoting immunity to pathogens, limiting inappropriate inflammatory responses, and mediating tissue repair. Our studies seek to gain insights into the underlying mechanisms of cancer development and identify new approaches for prevention and treatment.
Projects
Hereditary Cancer Syndromes
Hereditary gastrointestinal (GI) cancer syndromes are genetic disorders that predispose individuals to tumors in the GI tract and are also associated with extraintestinal manifestations. Examples for such syndromes are Lynch syndrome, familial adenomatous polyposis (FAP), MUTYH-associated polyposis, Peutz-Jeghers syndrome, juvenile polyposis, and Cowden disease. These conditions are caused by inherited mutations in various DNA repair or tumor-suppressor genes. Nonetheless, the presentation and clinical progression may differ among individuals carrying the same genetic variant, indicating that factors beyond genotype influence the disease. The local immune system is especially relevant in this regard, as numerous studies have demonstrated its role in shaping the development, progression, and treatment outcomes of various tumors. Consequently, we focus on understanding the composition and function of the intestinal immune infiltrate in these patients
Fibrosis
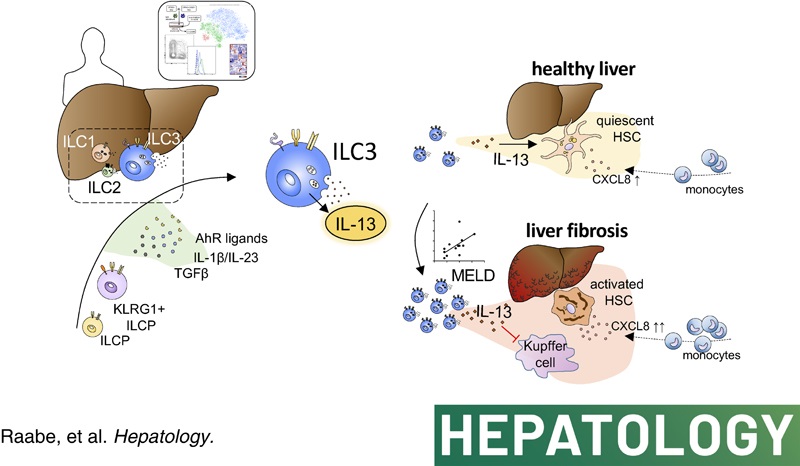
During the past years we could establish experimental evidence, that human NK cells also display anti-fibrotic activity via induction of hepatic stellate cell (HSC apoptosis). This anti-fibrotic function of cNK cells was linked to the surface expression of distinct molecules such as NKG2D and NKp46 and was importantly modified by factors from the microenvironment, such as neighbouring cells, the local cytokine milieu, and tissue oxygen concentrations. Recent data obtained in mouse models suggest that ILCs other than cNK cells also modulate hepatic fibrosis. Accordingly, current data of our group indicate that in human inflammatory liver disease both hepatic ILCs, via direct interaction with hepatic stellate cells, as well as intestinal ILCs, via modulating the gut-liver axis, modulate progression of liver fibrosis. Thus, it is the overall aim of the proposed project to analyse how liver disease affects compartment-specific composition and function of the ILC pool and to clarify how these alterations contribute to progression of liver fibrosis.
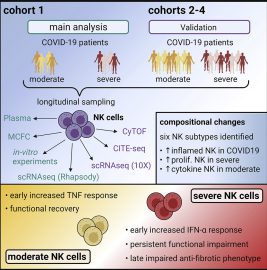
Viral Infections
Natural killer (NK) cells and innate lymphoid cells (ILCs) are important components of the innate immune system that play crucial roles in the immune response against viral infections. NK cells are a type of cytotoxic lymphocyte that can recognize and eliminate virally infected cells through a variety of mechanisms, including the release of cytotoxic granules, induction of apoptosis, and production of cytokines. Meanwhile, ILCs are a diverse population of lymphocytes that lack antigen-specific receptors but can rapidly produce cytokines in response to various stimuli, including viral infections. Our group is interested in the role of NK cells and ILCs in viral infections such as HIV and SARS-CoV2. Specifically, we are focused on investigating the impact of viral infections on NK cell function, the underlying mechanisms involved, and whether tissue-specific differences exist.Our group is interested in the role of NK cells and ILCs in viral infections such as HIV and SARS-CoV2. Specifically, we are focused on investigating the impact of viral infections on NK cell function, the underlying mechanisms involved, and whether tissue-specific differences exist.
Cell Rep. 2023 Jan 31;42(1):111937. doi: 10.1016/j.celrep.2022.11193
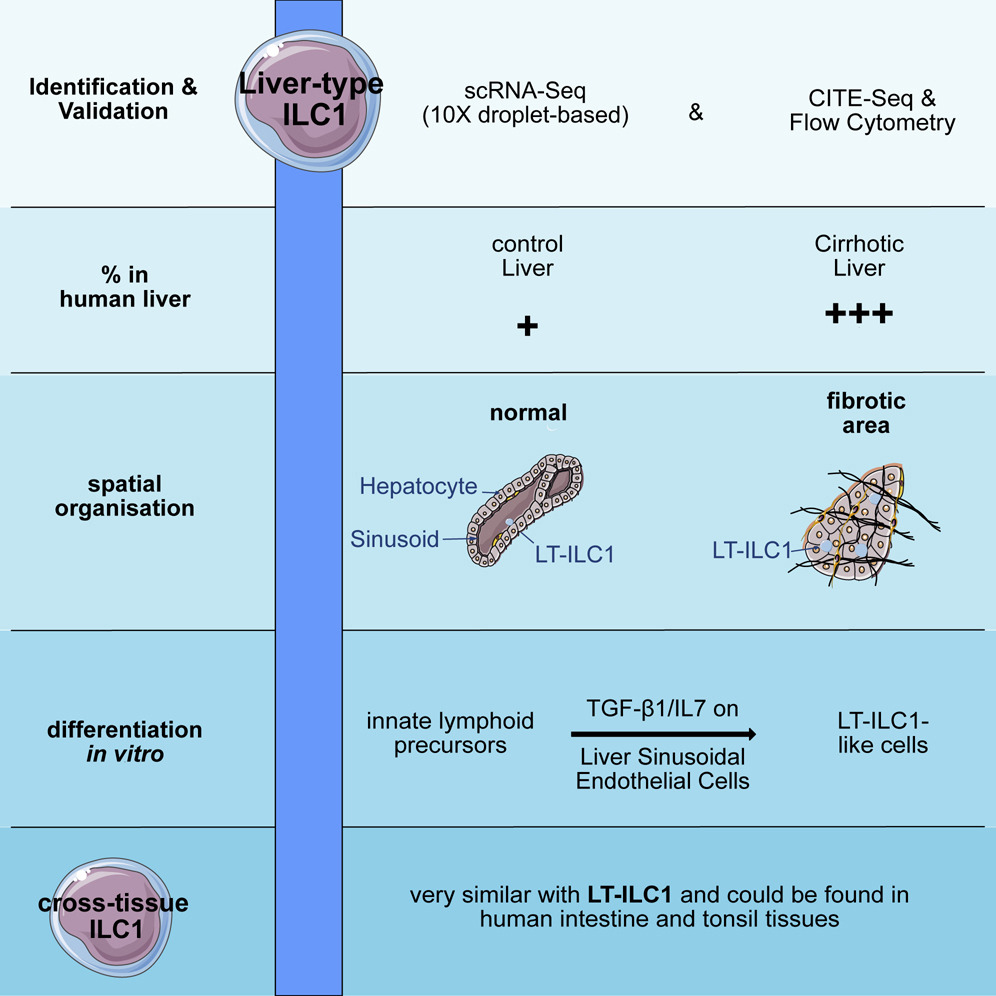
Hepatology ():10.1097/HEP.0000000000000350, April 10, 2023. | DOI: 10.1097/HEP.0000000000000350
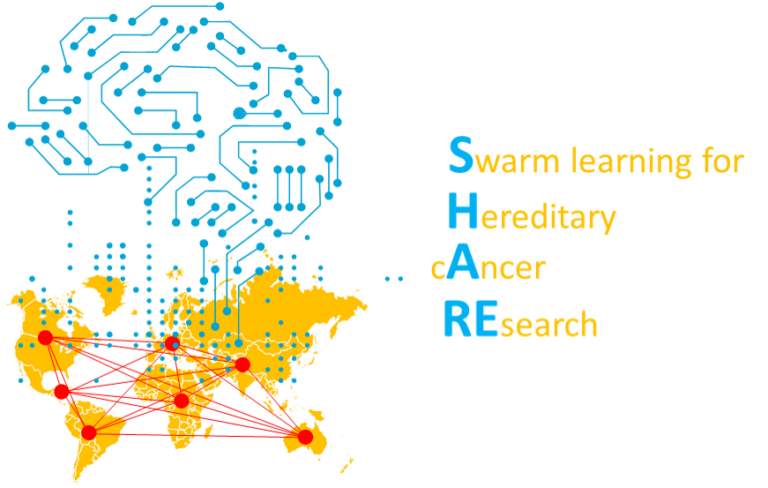
Our lab is partner of the SHARE initiative
Our team

Sofia Walferens
Federal Volunteer Service

Eyleen Majetec
MD student

Claudia Finnemann
Technician

Adrian Petras
MD student

Sandra Steinbach
MD student
Funding





Contact
Biomedical Center I (BMZ I)
Room 2OG006
Tel.: +49 (0) 228-287 -51416
Fax: +49 (0) 228-287 -51419